Regulatory Crackdown on Unlicensed IVF Devices in India as Fertility Industry Booms, ETHealthworld
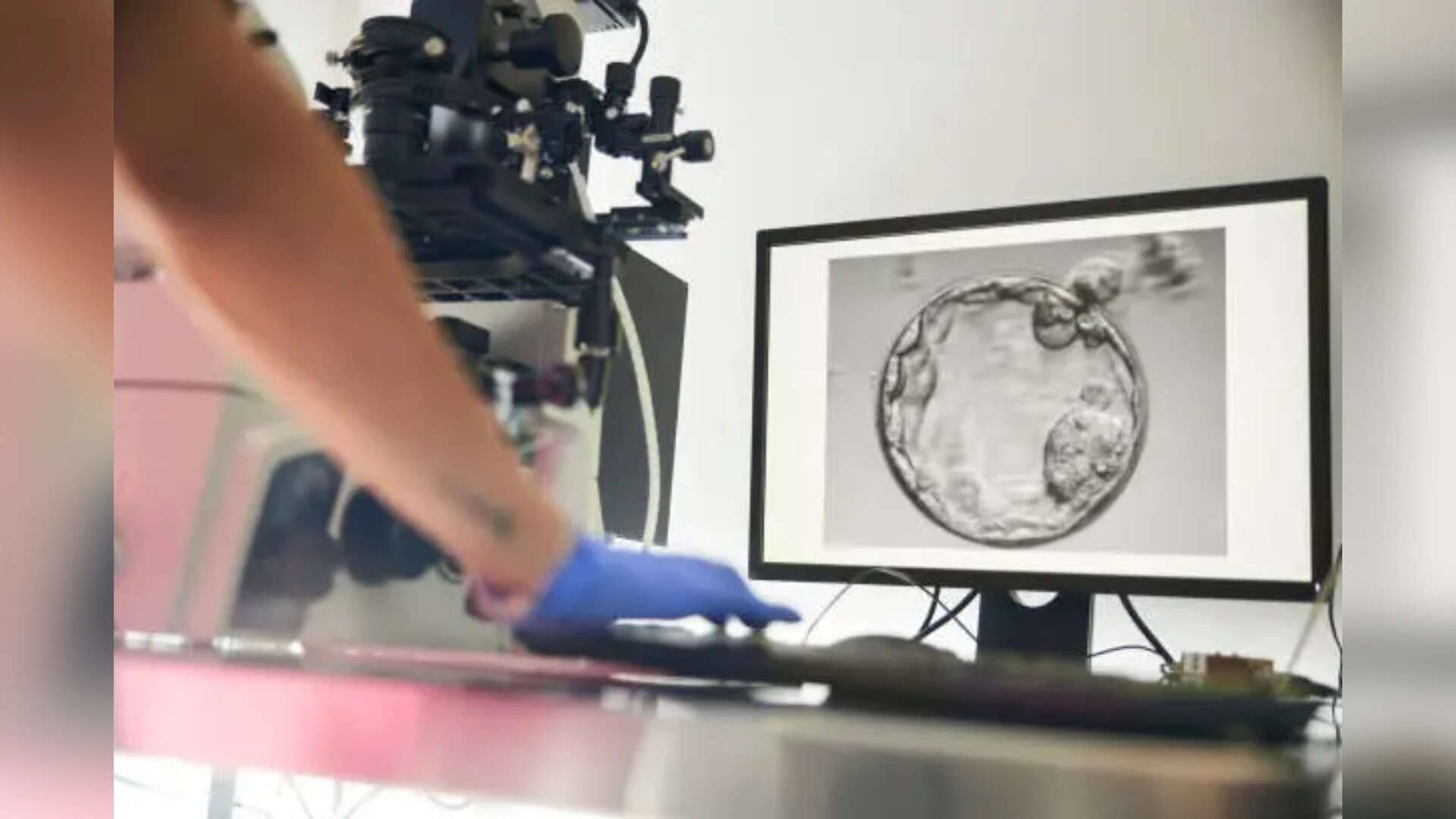
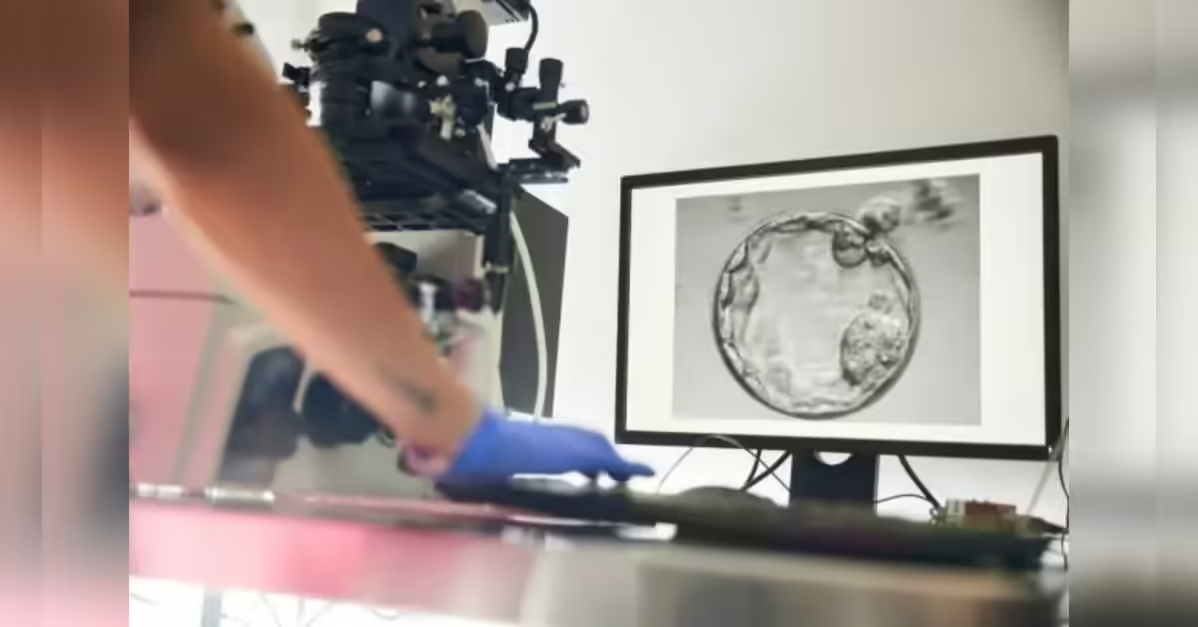
New Delhi: With India’s fertility industry expanding rapidly, Centre has turned its attention to a key blind spot – the devices used in IVF procedures – calling for stricter enforcement against their import, manufacture and sale.
A circular issued by the Central Drugs Standard Control Organisation (CDSCO) on Friday said devices used in assisted reproductive procedures, including intrauterine insemination (IUI) and sperm processing, were being sold in violation of regulatory norms.
The regulator made it clear that these products fell squarely within the definition of medical devices and were governed by the Drugs and Cosmetics Act, 1940, and the Medical Devices Rules, 2017. Under these provisions, licences are mandatory for import, manufacture, sale and distribution across the country.
At the same time, the rules provide a limited exemption for low-risk products – Class A devices that are non-sterile and non-measuring are kept outside the licensing requirement.
“It has been brought to the notice of this office that firms are selling medical devices intended for IVF… without obtaining licence,” the circular said, underlining concerns over compliance gaps.
Taking a serious view, the CDSCO has directed state drug controllers, licensing authorities, port offices and zonal regulators to maintain strict vigil.
Authorities have been asked to ensure that no unlicensed devices are imported, manufactured or allowed to circulate in the domestic market.
The move comes at a time when demand for assisted reproductive technologies is rising steadily, with more clinics and services coming up across urban centres.